– PDE9 inhibitor, CRD-740, demonstrated favorable safety profile and achieved statistical significance for the trial’s primary endpoint, median increase in plasma cyclic guanosine monophosphate (cGMP) –
– This clinical trial confirms that high levels of PDE9 inhibition lead to increases in cGMP, reflecting increased activation of the myocardial natriuretic peptide (NP) signaling pathway –
– Targeting PDE9 represents a novel approach to activating the NP signaling pathway, a highly validated pathway with established clinical benefits in heart failure –
– Clinical results presented today at the Annual Congress of the Heart Failure Association of the European Society of Cardiology –
BURLINGTON, Mass.–(BUSINESS WIRE)–Cardurion Pharmaceuticals, Inc. (“Cardurion”), a clinical-stage biotechnology company developing next-generation therapeutics for the treatment of cardiovascular diseases, today announced the presentation of positive clinical data from CARDINAL‑HF, the first Phase 2 proof-of-concept clinical trial of a phosphodiesterase-9 (PDE9) inhibitor in patients with heart failure. These data were presented today at the Annual Congress of the Heart Failure Association of the European Society of Cardiology taking place on May 11-14 in Lisbon, Portugal.

In the CARDINAL-HF Phase 2a trial, CRD-740 met the primary endpoint in patients with heart failure with reduced ejection fraction (HFrEF), demonstrating a statistically significant median increase in plasma cyclic guanosine monophosphate (cGMP) after four weeks of treatment. Plasma cGMP is a biomarker for intracellular cGMP whose levels reflect the activity of the protective myocardial natriuretic peptide (NP) signaling pathway, which has proven clinical benefits in heart failure. CRD-740 was generally well tolerated in the trial.
“These very promising data from the first Phase 2 proof-of-concept clinical trial of a PDE9 inhibitor in patients with heart failure represent an important next step in the development of this novel mechanism,” said James Udelson, MD, Chief of Cardiology at Tufts Medical Center and Principal Investigator for the CARDINAL-HF trial. “The results of this trial are impressive, showing robust PDE9 inhibition with significant increases in plasma and urinary cGMP, along with a favorable safety profile, both of which support moving into further clinical testing in heart failure.”
“Therapeutic targeting of the NP signaling pathway is precedented by today’s standard of care treatment for patients with heart failure, and PDE9 inhibition represents a new mechanism for activating this pathway to seek improved patient outcomes. Significant unmet needs remain, as heart failure is a prevalent and growing chronic condition that causes significant morbidity and mortality for millions of people,” said Scott D. Solomon, MD, Professor of Medicine at Harvard Medical School.
The oral presentation of these results, entitled, “A Phase 2, Randomized, Double-Blind, Placebo-Controlled Study to Assess the Tolerability and Pharmacodynamic Effects of CRD-740, a PDE9 Inhibitor, in Participants with Chronic Heart Failure,” were presented today by James Udelson, M.D., Principal Investigator of CARDINAL-HF and Chief of Cardiology at Tufts Medical Center, in the “Late-Breaking Clinical Trials: Medical Therapy” session at the Annual Congress of the Heart Failure Association of the European Society of Cardiology. Key findings presented include:
-
CRD-740 was generally well-tolerated in patients with HFrEF, a disease representing approximately one-half of patients with chronic heart failure.
-
PDE9 inhibition with CRD-740 achieved statistically significant median increases in plasma cGMP at four weeks, compared to placebo, which was the primary endpoint in the trial. Statistically significant median increases in urinary cGMP were also observed in the patients who received CRD-740, compared to placebo.
-
Increases in cGMP of the magnitude seen in the trial demonstrate that CRD-740 achieves high levels of PDE9 inhibition, which prevents cGMP metabolism by the PDE9 enzyme and leads to increased activation of the NP signaling pathway. Activation of the clinically validated NP signaling pathway has been shown to benefit patients with chronic heart failure in outcome studies with sacubitril/valsartan.
-
These increases in plasma and urinary cGMP were observed in patients who received CRD‑740 with and without background treatment with sacubitril/valsartan, supporting the potential for CRD-740 as a monotherapy, and as a new approach to augment efficacy in the setting of sacubitril/valsartan therapy and to further activate the NP signaling pathway.
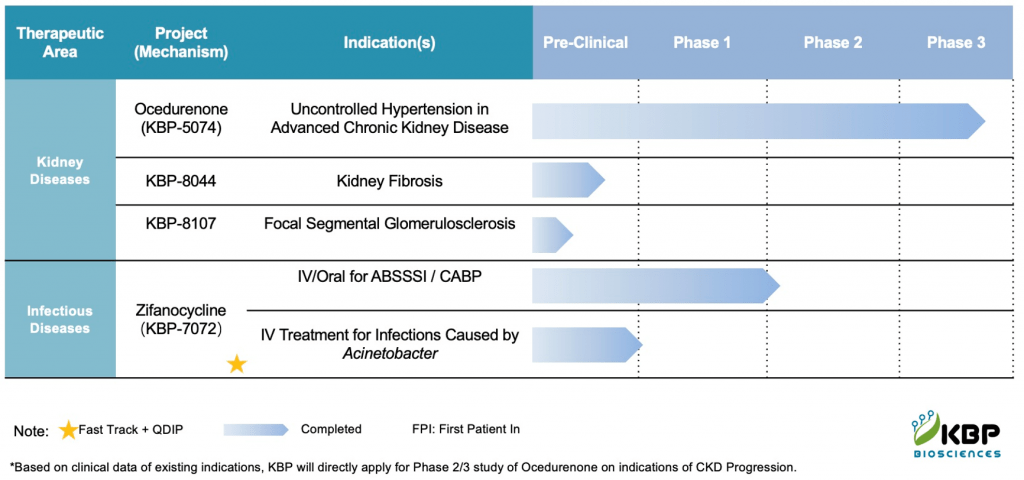
Based on the results of the CARDINAL-HF Phase 2a trial, Cardurion has launched two Phase 2 clinical trials in 640 patients, including a dose-ranging trial in patients with HFrEF and a proof-of-concept trial in patients with heart failure with preserved ejection fraction (HFpEF).
“These findings from Cardurion’s pioneering program in PDE9 support our conviction that PDE9 inhibition presents an important new mechanism for independently targeting and further activating the well-validated NP signaling pathway. The data from this trial suggest that PDE9 inhibition has the potential to provide benefit to patients when administered alone or in combination with guideline directed medical therapy, and ultimately become standard of care for patients with both types of heart failure,” said Peter Lawrence, Chief Executive Officer of Cardurion Pharmaceuticals.
“We are delighted to share these clinical results for CRD-740 as our team advances PDE9 inhibition as a novel approach to addressing the unmet needs of patients with chronic heart failure,” said Howard Surks, MD, Chief Medical and Scientific Officer of Cardurion Pharmaceuticals. “We thank our patients and investigators for their partnership and look forward to continuing the development of our PDE9 inhibitors to improve outcomes for patients.”
About the CARDINAL-HF clinical trial
CARDINAL-HF is the first Phase 2 proof-of-concept trial of a PDE9 inhibitor for the treatment of patients with heart failure. The Phase 2a clinical trial is a randomized, placebo-controlled trial of 60 chronic, stable patients with heart failure with reduced ejection fraction (HFrEF) who were receiving guideline-directed medical therapy (GDMT). The primary endpoints of the trial are safety and tolerability of CRD-740 and changes in plasma cGMP at four weeks, a precedented biomarker for activation of the NP signaling pathway. Other endpoints include changes in urinary cGMP and N-terminal pro b-type natriuretic peptide (NTpro-BNP) and effect of CRD-740 administration on the Kansas City Cardiomyopathy questionnaire (KCCQ), which measures symptoms, physical and social limitations, and quality of life in patients with heart failure.
The Executive Committee for the CARDINAL-HF trial includes Dr. James Udelson, Principal Investigator and Chief of Cardiology at Tufts Medical Center; Dr. Scott Solomon, Professor of Medicine at Harvard Medical School; and Dr. John McMurray, Professor of Medical Cardiology and Deputy Director of the Institute of Cardiovascular and Medical Sciences at the University of Glasgow. Drs. Udelson, Solomon and McMurray, along with Dr. Eugene Braunwald, the Distinguished Hersey Professor of Medicine at Harvard Medical School, also lead Cardurion’s Clinical Advisory Board. The CARDINAL-HF Steering Committee is further comprised of leading international heart failure clinical investigators.
About PDE9 inhibition
PDE9 inhibitors target phosphodiesterase (PDE) 9, an enzyme whose elevated activity in patients with heart failure reduces the beneficial effects of the natriuretic peptide (NP) pathway, fundamental to cardiovascular homeostasis. The beneficial effects of the NP signaling pathway in the heart muscle cell are mediated by cyclic guanosine monophosphate (cGMP) and activation of its downstream signaling molecule protein kinase G1. PDE9 is an enzyme that selectively degrades cGMP; therefore, by inhibiting PDE9, our goal is to preserve cGMP generation and enhance the beneficial natriuretic peptide signaling within heart muscle cells.
About Chronic Heart Failure
Heart failure is a prevalent and growing condition that is responsible for substantial morbidity and mortality. Approximately 6.5 million people in the United States suffer from heart failure, and 50 percent of these patients die from the condition within five years of diagnosis. One in five patients with heart failure are hospitalized annually, and 25% of these patients will be admitted again within a month of discharge. Approximately half of all patients with heart failure have reduced ejection fraction (HFrEF) and half have preserved ejection fraction (HFpEF). Despite the availability of drugs indicated to reduce morbidity and mortality in patients with both types of heart failure, substantial unmet medical need remains.
About Cardurion Pharmaceuticals
Cardurion Pharmaceuticals is a clinical-stage biotechnology company focused on the discovery and development of novel, next-generation therapeutics for the treatment of cardiovascular diseases. Cardurion was founded by physician-scientists with world-class expertise in cardiovascular signaling pathways, and a shared passion to find and develop a pipeline of novel treatment options to improve the lives of patients. Cardurion has two groundbreaking clinical programs in development, a PDE9 inhibitor targeting heart failure and the first ever CaMKII inhibitor in clinical development targeting multiple cardiovascular indications.
Cardurion Pharmaceuticals has facilities in Burlington, Massachusetts and Shonan, Japan. For more information, please visit the company’s website at https://cardurion.com.
Contacts
Kathryn Morris
The Yates Network
kathryn@theyatesnetwork.com